The nucleus of the most common isotope of the hydrogen atom (with the chemical symbol "H") is a lone proton. Protons and neutrons are both nucleons, which may be bound together by the nuclear force to form atomic nuclei. Protons have a positive charge distribution, which decays approximately exponentially, with a root mean square charge radius of about 0.8 fm. : 21–22 A modern perspective has a proton composed of the valence quarks (up, up, down), the gluons, and transitory pairs of sea quarks. The two up quarks and one down quark of a proton are held together by the strong force, mediated by gluons. Protons are spin- 1 / 2 fermions and are composed of three valence quarks, making them baryons (a sub-type of hadrons). When free hydrogen atoms react with each other, they form neutral hydrogen molecules (H 2), which are the most common molecular component of molecular clouds in interstellar space.įree protons are routinely used for accelerators for proton therapy or various particle physics experiments, with the most powerful example being the Large Hadron Collider. Such "free hydrogen atoms" tend to react chemically with many other types of atoms at sufficiently low energies. In a vacuum, when free electrons are present, a sufficiently slow proton may pick up a single free electron, becoming a neutral hydrogen atom, which is chemically a free radical. 
The result is a protonated atom, which is a chemical compound of hydrogen. A fast proton moving through matter will slow by interactions with electrons and nuclei, until it is captured by the electron cloud of an atom. However, the character of such bound protons does not change, and they remain protons. At sufficiently low temperatures and kinetic energies, free protons will bind to electrons. įree protons occur occasionally on Earth: thunderstorms can produce protons with energies of up to several tens of MeV. In 2019, two different studies, using different techniques, found this radius to be 0.833 fm, with an uncertainty of ☐.010 fm. Because protons are not fundamental particles, they possess a measurable size the root mean square charge radius of a proton is about 0.84–0.87 fm ( 1 fm = 10 −15 m). The remainder of a proton's mass is due to quantum chromodynamics binding energy, which includes the kinetic energy of the quarks and the energy of the gluon fields that bind the quarks together. The rest masses of quarks contribute only about 1% of a proton's mass. Protons are composed of two up quarks of charge + 2 / 3 e and one down quark of charge − 1 / 3 e. 
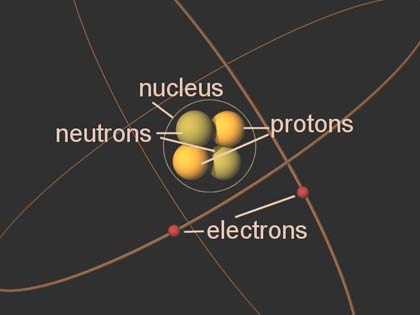
Protons were therefore a candidate to be a fundamental or elementary particle, and hence a building block of nitrogen and all other heavier atomic nuclei.Īlthough protons were originally considered to be elementary particles, in the modern Standard Model of particle physics, protons are now known to be composite particles, containing three valence quarks, and together with neutrons are now classified as hadrons. In previous years, Rutherford had discovered that the hydrogen nucleus (known to be the lightest nucleus) could be extracted from the nuclei of nitrogen by atomic collisions. The word proton is Greek for "first", and this name was given to the hydrogen nucleus by Ernest Rutherford in 1920. Since each element has a unique number of protons, each element has its own unique atomic number, which determines the number of atomic electrons and consequently the chemical characteristics of the element. The number of protons in the nucleus is the defining property of an element, and is referred to as the atomic number (represented by the symbol Z). They provide the attractive electrostatic central force that binds the atomic electrons. One or more protons are present in the nucleus of every atom. Protons and neutrons, each with masses of approximately one atomic mass unit, are jointly referred to as " nucleons" (particles present in atomic nuclei). Its mass is slightly less than that of a neutron and 1,836 times the mass of an electron (the proton-to-electron mass ratio). , H +, or 1H + with a positive electric charge of +1 e ( elementary charge). Identified in other nuclei (and named) by Ernest Rutherford (1917–1920).Ī proton is a stable subatomic particle, symbol Observed as H + by Eugen Goldstein (1886). Forces between quarks are mediated by gluons. The color assignment of individual quarks is arbitrary, but all three colors must be present. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
